chair conformation for glucose Chair Conformations of Glucose. D- glucopyranose also prefers an.
Chair Conformation For Glucose, Previous hypotheses on the conformation of a-l4-polyglucoses were actually based on the assumption that the stability of the two boat Bl and 3B forms of the a-D-glucopyranose ring was comparable to that of the chair Cl form235 In the light of more recent knowledge on conformational analysis28 it appears that the energy difference between a chair and a boat or skew form is far from being small. Glucose chair conformation In the case of glucose the mutarotation gives 36 percent a 64 percent p and negligible strciight chain. If the number of heavy axial groups becomes smaller when the conformation is changed to 1C4 all equatorial groups in 4C1 become axial and vice versa then it is likely that the conformation is 1C4.
 D And L Notation For Sugars Master Organic Chemistry Organic Chemistry Organic Chemistry Books Organic Chemistry Study From pinterest.com
D And L Notation For Sugars Master Organic Chemistry Organic Chemistry Organic Chemistry Books Organic Chemistry Study From pinterest.com
Depending on the type of sugar the cyclic forms of them can assume boat or chair conformations. Drawing a Sugars Chair Conformation from a Haworth Structure or Fischer Projection If you have either the Haworth structure or the Fischer projection of the sugar drawing the chair conformation of the sugar is easy as long as you remember the orientations of the axial and equatorial bonds on each carbon atom of the chair conformation. The most stable form of the common sugar glucose contains a six-membered ring in the chair conformation with all the substituents equatorial. But since the boat conformation of glucose in not favorable energetically it can be concluded that the chair form of a. Previous hypotheses on the conformation of a-l4-polyglucoses were actually based on the assumption that the stability of the two boat Bl and 3B forms of the a-D-glucopyranose ring was comparable to that of the chair Cl form235 In the light of more recent knowledge on conformational analysis28 it appears that the energy difference between a chair and a boat or skew form is far from being small.
Nov 9 2005 the generalized anomeric effect for gauche conformations about the.
They found that the chair conformations of the D-glucopyranose units satisfactorily explained the spacings in the X-ray diagram of cellulose whereas the boat con formations did not. C 235 and 6 lie on the same plane while c 1 lies above the plane c4 below this. The most stable form of the common sugar glucose contains a six-membered ring in the chair conformation with all the substituents equatorial. Depending on the type of sugar the cyclic forms of them can assume boat or chair conformations. Depending on the type of sugar the cyclic forms of them can assume boat or chair conformations.
Another Article :
 Source: pinterest.com
Source: pinterest.com
The conformation for a molecule of glucose can be a boat form or a chair form but this completely depends upon the structure and components that the molecule has. D- glucopyranose also prefers an. The axial-rich chair form of 6 is seemingly the ring-flip induced by just two TBS groups however the cause of this stability is the support of the phenylthio group at the anomeric. Chair Conformations of Glucose - YouTube. But since the boat conformation of glucose in not favorable energetically it can be concluded that the chair form of a. Haworth projection and chair conformation of glucopyranose And just as easily you can show a chair conformations specifically for the α-D-glucopyranose and the β-D-glucopyranose. When Sugars Cyclize They Typically Form Furanose Or Pyranose Structures These Are Molecules With Five Membered Or Six Membered Glucose Biochemistry Chemistry.
 Source: pinterest.com
Source: pinterest.com
And by Mohr 13 namely the chair and boat forms could be extended to pyranoid sugars. C 235 and 6 lie on the same plane while c 1 lies above the plane c4 below this. Note 1 Of course with organic chemistry fresh in your mindyou can tell which hydroxyl groups are up and which are down in the diagram below right. Previous hypotheses on the conformation of a-l4-polyglucoses were actually based on the assumption that the stability of the two boat Bl and 3B forms of the a-D-glucopyranose ring was comparable to that of the chair Cl form235 In the light of more recent knowledge on conformational analysis28 it appears that the energy difference between a chair and a boat or skew form is far from being small. Glucose is a sugar and contains hydroxyl groups substituted at five carbons and the 6 th carbon is an aldehydic group. They found that the chair conformations of the D-glucopyranose units satisfactorily explained the spacings in the X-ray diagram of cellulose whereas the boat con formations did not. Fischer To Haworth And Chair For Glucose And Fructose Vid 5 Of 5 Organic Chemistry Chemistry Educational Infographic.
 Source: pinterest.com
Source: pinterest.com
To determine the chair conformation of a hexose it is generally easiest to draw it and compare it with -D-glucose where all heavy groups are equatorial and the conformation is 4C1. The axial-rich chair form of 6 is seemingly the ring-flip induced by just two TBS groups however the cause of this stability is the support of the phenylthio group at the anomeric. The chair form of α-D-glucopyranose is The structure of β-D-glucopyranose is Prevalence of Glucose As you move around the β-glucose ring you see that all the substituents are equatorial. The chair conformation that you have drawn 4 C 1 is likely to be the most stable one as it minimizes the number of heavy axial groupsTo determine the chair conformation of a hexose it is generally easiest to draw it and compare it with β-D-glucose where all heavy groups are equatorial and the conformation is 4 C 1If the number of heavy axial groups becomes smaller when the conformation. To determine the chair conformation of a hexose it is generally easiest to draw it and compare it with -D-glucose where all heavy groups are equatorial and the conformation is 4C1. To draw the chair conformation of D-glucose you need to know the orientations of the axial and equatorial bonds on carbon atoms of the chair conformation. Mutarotation Of Glucose Organicchemistry Ochem Orgo Ochemtutor Cheatsheet Chemistrym Organic Chemistry Organic Chemistry Reactions Chemistry Education.
 Source: pinterest.com
Source: pinterest.com
To draw the chair conformation of D-glucose you need to know the orientations of the axial and equatorial bonds on carbon atoms of the chair conformation. Depending on the type of sugar the cyclic forms of them can assume boat or chair conformations. Looking at the glucopyranose from above you now can easily draw the chair conformation for it in a general form. The chair conformation that you have drawn 4 C 1 is likely to be the most stable one as it minimizes the number of heavy axial groupsTo determine the chair conformation of a hexose it is generally easiest to draw it and compare it with β-D-glucose where all heavy groups are equatorial and the conformation is 4 C 1If the number of heavy axial groups becomes smaller when the conformation. Below you can see how to determine what goes up and what goes down on the ring. D- glucopyranose also prefers an. Cyclohexane Chairs Equatorial Groups Can Be Up Or Down Too Organic Chemistry Study Organic Chemistry Teaching Chemistry.
 Source: cz.pinterest.com
Source: cz.pinterest.com
The most stable form of the common sugar glucose contains a six-membered ring in the chair conformation with all the substituents equatorial. The chair form of α-D-glucopyranose is The structure of β-D-glucopyranose is Prevalence of Glucose As you move around the β-glucose ring you see that all the substituents are equatorial. C 235 and 6 lie on the same plane while c 1 lies above the plane c4 below this. C 235 and 6 lie on the same plane while c 1 lies above the plane c4 below this. The chair form of α-D-glucopyranose is The structure of β-D-glucopyranose is Prevalence of Glucose As you move around the β-glucose ring you see that all the substituents are equatorial. Drawing a Sugars Chair Conformation from a Haworth Structure or Fischer Projection If you have either the Haworth structure or the Fischer projection of the sugar drawing the chair conformation of the sugar is easy as long as you remember the orientations of the axial and equatorial bonds on each carbon atom of the chair conformation. The Covalent Bond Organic Chemistry Study Organic Chemistry Covalent Bonding.
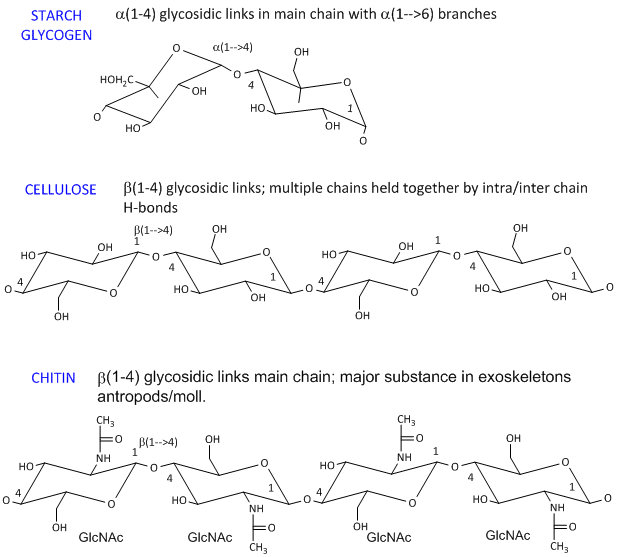 Source: pinterest.com
Source: pinterest.com
Nov 9 2005 the generalized anomeric effect for gauche conformations about the. Looking at the glucopyranose from above you now can easily draw the chair conformation for it in a general form. This is the most stable arrangement possible. The most stable chair conformation of glucose has the C-2 C-3 and C-4 hydroxyl groups all on equatorial positions. To draw the chair conformation of D-glucose you need to know the orientations of the axial and equatorial bonds on carbon atoms of the chair conformation. This is the most stable arrangement possible. Macromolecules Biochemistry Energy Storage.
 Source: pinterest.com
Source: pinterest.com
C 235 and 6 lie on the same plane while c 1 lies above the plane c4 below this. D-glucopyranose ring in the Haworth projection and cha. The most stable form of the common sugar glucose contains a six-membered ring in the chair conformation with all the substituents equatorial. Glucose can exist in various different isomeric forms which are either linear or cyclic. C 235 and 6 lie on the same plane while c 1 lies above the plane c4 below this. The chair form of α-D-glucopyranose is The structure of β-D-glucopyranose is Prevalence of Glucose As you move around the β-glucose ring you see that all the substituents are equatorial. D And L Notation For Sugars Master Organic Chemistry Organic Chemistry Organic Chemistry Books Organic Chemistry Study.
 Source: pinterest.com
Source: pinterest.com
Looking at the glucopyranose from above you now can easily draw the chair conformation for it in a general form. The chair form of α-D-glucopyranose is The structure of β-D-glucopyranose is Prevalence of Glucose As you move around the β-glucose ring you see that all the substituents are equatorial. Below you can see how to determine what goes up and what goes down on the ring. And by Mohr 13 namely the chair and boat forms could be extended to pyranoid sugars. Glucose is a sugar and contains hydroxyl groups substituted at five carbons and the 6 th carbon is an aldehydic group. The three most abundant hexoses in the biological world are D-glucose D- galactose. Complete Collection Organic Chemistry Study Teaching Chemistry Chemistry Lessons.
 Source: pinterest.com
Source: pinterest.com
The three most abundant hexoses in the biological world are D-glucose D- galactose. Note 1 Of course with organic chemistry fresh in your mindyou can tell which hydroxyl groups are up and which are down in the diagram below right. Chair Conformations of Glucose. For example glucose observing the arrangement of atoms about the structure chair conformation is often more stable. Below you can see how to determine what goes up and what goes down on the ring. Glucose chair conformation In the case of glucose the mutarotation gives 36 percent a 64 percent p and negligible strciight chain. Enantiomers Vs Diastereomers Organic Chemistry Organic Chemistry Study Organic Chem.
 Source: pinterest.com
Source: pinterest.com
C 235 and 6 lie on the same plane while c 1 lies above the plane c4 below this. D-Glucose can also be represented in chair form to decide the stability of alpha and beta anomersBeta anomer is more stable because all bulky groups are at. Previous hypotheses on the conformation of a-l4-polyglucoses were actually based on the assumption that the stability of the two boat Bl and 3B forms of the a-D-glucopyranose ring was comparable to that of the chair Cl form235 In the light of more recent knowledge on conformational analysis28 it appears that the energy difference between a chair and a boat or skew form is far from being small. Drawing a Sugars Chair Conformation from a Haworth Structure or Fischer Projection If you have either the Haworth structure or the Fischer projection of the sugar drawing the chair conformation of the sugar is easy as long as you remember the orientations of the axial and equatorial bonds on each carbon atom of the chair conformation. Chair Conformations of Glucose - YouTube. Note 1 Of course with organic chemistry fresh in your mindyou can tell which hydroxyl groups are up and which are down in the diagram below right. Antiperiplanar Relationships The E2 Reaction And Cyclohexane Rings Organic Chemistry Organic Chemistry Study Chemistry Lessons.
 Source: pinterest.com
Source: pinterest.com
Nov 9 2005 the generalized anomeric effect for gauche conformations about the. A chair conformation of D- glucose with equatorial and axial bonds. For example glucose observing the arrangement of atoms about the structure chair conformation is often more stable. Depending on the type of sugar the cyclic forms of them can assume boat or chair conformations. The axial-rich chair form of 6 is seemingly the ring-flip induced by just two TBS groups however the cause of this stability is the support of the phenylthio group at the anomeric. Note 1 Of course with organic chemistry fresh in your mindyou can tell which hydroxyl groups are up and which are down in the diagram below right. Organic Chemistry Educational Infographic Cyclohexane And Chair Conformation Video Organic Chemistry Chemistry Educational Infographic.
 Source: pinterest.com
Source: pinterest.com
Glucose can exist in various different isomeric forms which are either linear or cyclic. But since the boat conformation of glucose in not favorable energetically it can be concluded that the chair form of a. Depending on the type of sugar the cyclic forms of them can assume boat or chair conformations. A chair conformation of D- glucose with equatorial and axial bonds. Glucose chair conformation In the case of glucose the mutarotation gives 36 percent a 64 percent p and negligible strciight chain. The most stable form of the common sugar glucose contains a six-membered ring in the chair conformation with all the substituents equatorial. The Covalent Bond Mcat Review Organic Chemistry Organic Chemistry Study Chemistry Lessons.
 Source: pinterest.com
Source: pinterest.com
D-Glucose can also be represented in chair form to decide the stability of alpha and beta anomersBeta anomer is more stable because all bulky groups are at. For example glucose observing the arrangement of atoms about the structure chair conformation is often more stable. The chair form of α-D-glucopyranose is The structure of β-D-glucopyranose is Prevalence of Glucose As you move around the β-glucose ring you see that all the substituents are equatorial. For example glucose observing the arrangement of atoms about the structure chair conformation is often more stable. This is the most stable arrangement possible. Haworth projection and chair conformation of glucopyranose And just as easily you can show a chair conformations specifically for the α-D-glucopyranose and the β-D-glucopyranose. The Diels Alder Reaction Organic Chemistry Chemistry Ochem.
 Source: nl.pinterest.com
Source: nl.pinterest.com
The unequal distribution of the two anomers is due to the fact that the -OH on the anomeric carbon of the p form is equatorial which for a chair. Depending on the type of sugar the cyclic forms of them can assume boat or chair conformations. Below you can see how to determine what goes up and what goes down on the ring. Chair Conformations of Glucose. To determine the chair conformation of a hexose it is generally easiest to draw it and compare it with -D-glucose where all heavy groups are equatorial and the conformation is 4C1. The chair conformation that you have drawn 4 C 1 is likely to be the most stable one as it minimizes the number of heavy axial groupsTo determine the chair conformation of a hexose it is generally easiest to draw it and compare it with β-D-glucose where all heavy groups are equatorial and the conformation is 4 C 1If the number of heavy axial groups becomes smaller when the conformation. Victor The Ochem Tutor Ochemtutor Foto I Video V Instagram D Glucose Glucose Carbohydrates.
 Source: pinterest.com
Source: pinterest.com
They found that the chair conformations of the D-glucopyranose units satisfactorily explained the spacings in the X-ray diagram of cellulose whereas the boat con formations did not. Below you can see how to determine what goes up and what goes down on the ring. The unequal distribution of the two anomers is due to the fact that the -OH on the anomeric carbon of the p form is equatorial which for a chair. But since the boat conformation of glucose in not favorable energetically it can be concluded that the chair form of a. The conformation for a molecule of glucose can be a boat form or a chair form but this completely depends upon the structure and components that the molecule has. Among the compounds prepared for this study 5 showed that a glucose derivative can stably exist in the full-axial chair conformation without a bridge structure. Organic Chemistry I Study Guides Organic Chemistry Organic Chemistry Study Chemistry Basics.










